What Is REGENCOV Antibody: New COVID-19 Treatment, Effectiveness, Dosage
 |
| What Is REGEN-COV Antibody: New COVID-19 Treatment - Photo: News Medical |
Regeneron's REGEN-COV Antibody Cocktail is a combination of monoclonal antibodies casirivimab and imdevimab, explicitly designed to block the infectivity of SARS-CoV-2, the coronavirus that causes COVID-19. This was the first antibody therapy to demonstrate an anti-viral effect in patients hospitalized with COVID-19.
Regeneron's core technologies allow for the rapid and efficient generation of these protective anti-viral antibodies outside of the body, derived from genetically humanized mice or convalescent humans. The resulting antibodies correspond to the most potent anti-viral antibodies that could be elicited by a vaccine or through exposure to a pathogen.
These REGEN-COV antibodies can be delivered to people via injection, providing "passive immunity" and immediately protecting the disease. However, they must be re-administered to remain effective over time. These antibodies can also treat an existing infection, unlike vaccines, which can only be used preventatively.
History of REGEN-COV Antibody
The concept that drug cocktails can prevent viral escape has previously been demonstrated for traditional antiviral drugs used to treat HIV and other viruses. However, Regeneron now reports the fundamental realization that this can also be true for antibody-based therapies, as reported in a paper published in Science on August 21, 2020, entitled "Antibody Cocktail to SARS-Cov-2 Spike Protein Prevents Rapid Mutational Escape Seen with Individual Antibodies," which further defines the protective value of the multiple-antibody approach against SARS-CoV-2 specifically.
Regeneron scientists evaluated thousands of fully human antibodies produced by the company's proprietary VelocImmune® mice, which have been genetically modified to have a human immune system, as well as antibodies isolated from humans who have recovered from COVID-19.
Regeneron has recently partnered with Roche on August 19, 2020, to increase COVID-19 Antibody Cocktail's global supply. If COVID-19 Antibody Cocktail proves safe and effective in clinical trials and regulatory approvals are granted, Regeneron will manufacture and distribute it in the U.S., and Roche will develop, manufacture, and distribute it outside the USA.
 |
| Study: Subcutaneous REGEN-COV Antibody Combination in Early SARS-CoV-2 Infection. Image Credit: NIAID |
On October 7, 2020, the company issued a 'Statement on REGN-COV2 Emergency Use Authorization Request: After our discussions with regulatory authorities, we have submitted a request to the U.S. Food and Drug Administration for an Emergency Use Authorization (EUA) for our COVID-19 Antibody Cocktail investigational antibody combination for COVID-19. Under our agreement with the U.S. government for the initial doses of COVID-19 Antibody Cocktail, if a EUA is granted, the government has committed to making these doses available to the American people at no cost and would be responsible for their distribution. At this time, there are doses available for approximately 50,000 patients, and we expect to have doses available for 300,000 patients in total within the next few months.'
On December 29, 2020, the company announced 'the Phase 3 program in hospitalized patients to continue based on passing futility analysis on the ability to reduce the incidence of death or mechanical ventilation. As in an earlier outpatient trial, immune status when patients entered the trial was a strong predictor of viral load and clinical outcomes.' "In this trial, patients who had not yet mounted their own immune response had much higher viral loads and much worse clinical outcomes; for example, in the placebo group, seronegative patients were almost three times more likely to die compared to seropositive patients," stated George D. Yancopoulos, M.D., Ph.D., President, and Chief Scientific Officer at Regeneron.
This COVID-19 Antibody Cocktail's development and manufacturing has been funded in part with federal funds from the Biomedical Advanced Research and Development Authority under OT number: HHSO100201700020C.
New York-based Regeneron (REGN) is a leading biotechnology company that invents life-transforming medicines for people with serious diseases.
Effectiveness of REGEN-COV Antibody
Researchers in the United States have demonstrated the effectiveness of early treatment with REGEN-COV at preventing symptomatic disease among people infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) – the agent that causes coronavirus disease 2019 (COVID-19).
The antibody combination treatment has already been shown to be effective at reducing the risk of hospitalization and death among individuals with symptomatic COVID-19.
Now, Meagan O’Brien from the University of Pennsylvania in Philadelphia and colleagues have shown that individuals infected with SARS-CoV-2 who have not yet developed any symptoms also benefit from receiving REGEN-COV, with the treatment reducing the risk of symptomatic disease by 31.5%.
The team suggests that using REGEN-COV as a complement to vaccination may dramatically reduce the overall reservoir of virus in the community and decrease disease severity and healthcare utilization among infected individuals.
REGEN-COV comprises two neutralizing monoclonal antibodies – casirivimab and imdevimab – that bind distinct epitopes on the SARS-CoV-2 spike protein receptor-binding domain (RBD) and block virus entry into host cells. The spike RBD initiates the initial stage of the infection process when it binds to the human host cell receptor angiotensin-converting enzyme 2 (ACE2).
Intravenous administration of this two-antibody combination has already been shown to significantly reduce the risk of hospitalization and death in outpatients with symptomatic COVID-19.
REGENCOV has an acceptable safety profile
Among those who received REGEN-COV, no COVID-19-related hospitalization or emergency room (ER) visits occurred. In contrast, three placebo recipients visited ER, one was hospitalized, and two visited ER and were subsequently hospitalized.
The proportion of participants who had one or more treatment-emergent adverse events was 48.1% among placebo recipients, compared with 33.5% for those receiving REGEN-COV.
What did the current study involve?
For the study, 314 individuals (aged 12 years or more) who were recently infected with SARS-CoV-2 through contact with a household index case, but had not yet developed any symptoms, were randomly assigned to receive a single dose of REGEN-COV 1200mg or placebo by subcutaneous injection.
The primary endpoint was the proportion of infected participants without evidence of previous immunity who subsequently developed symptomatic COVID-19 during a 28-day efficacy assessment.
The trial took place at 112 sites in the United States, Romania, and Moldova. It was managed jointly by Regeneron, the COVID-19 Prevention Network, and the National Institute of Allergy and Infectious Diseases.
Dosage
On June 3, 2021, the U.S. Food and Drug Administration updated the Emergency Use Authorization for REGEN-COV, lowering the dose to 1,200 mg (600 mg casirivimab and 600 mg imdevimab) is half the dose originally authorized. As part of the updated EUA, REGEN-COV should be administered by intravenous infusion; subcutaneous injections are an alternative when IV infusion is not feasible and would lead to a delay in treatment.
Healthcare providers should review the Fact Sheet for Healthcare Providers for information on the authorized use of REGEN-COV and mandatory requirements of the EUA and comply with the requirements of the EUA.
| AN ANTIBODY THERAPY from Regeneron, a firm in upstate New York, improves the survival of patients with covid-19 and offers renewed hope for the treatment of those most seriously ill with the disease. A study in British hospitals found that Regen-Cov saved the lives of many of those unable to make their own antibodies in response to SARS-CoV-2. Such “seronegative” individuals constituted about a third of the 9,785 hospital patients in the study. As compared with a control group given standard treatment (either a steroid called dexamethasone or, for the sickest, an anti-inflammatory known as tocilizumab), 20% more patients survived. Regen-Cov also reduced the median length of hospital stays from 17 to 13 days. Regen-Cov is a combination of two monoclonal antibodies, known as casirivimab and imdevimab. Antibodies are immune-system proteins that disable pathogens by locking specifically onto them. Both casirivimab and imdevimab bind to different sites on the coronavirus’s “spike” protein, preventing the virus from infecting cells. Using two antibodies instead of one reduces the risk of the virus evolving resistance to the treatment. |
 Top 9 Countries with the Most COVID-19 Vaccinations and The Ranking of Countries Per Doses Top 9 Countries with the Most COVID-19 Vaccinations and The Ranking of Countries Per Doses More than 2 billion COVID-19 vaccines have now been given out across the world. Check out The Top 9 Countries with the Most COVID-19 Vaccinations ... |
 Top 9 Biggest Vaccine Companies In The World Top 9 Biggest Vaccine Companies In The World Several leading vaccine manufacturers are spending heavily to develop vaccination for babies and vaccination schedule for adults in the wake of rising number of diseases ... |
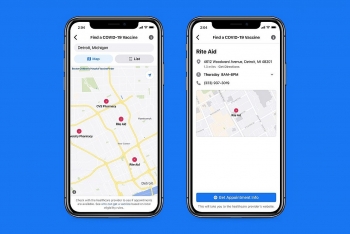 Facebook vaccine finder is launching to help you get your Covid vaccine appointment Facebook vaccine finder is launching to help you get your Covid vaccine appointment Facebook announced Monday the launch of its vaccine finder tool to help find where and when Rio Grande Valley residents can get a COVID-19 vaccine. ... |
 What is COVID-19 or Coronavirus? What is COVID-19 or Coronavirus? COVID-19 is a dangerous disease that the whole world is facing. So, follow the article below to find out what it is and how it ... |