What are the Rarest Elements On Earth - Top 11
 |
| Top Rarest Elements On Earth Today. Photo: KnowInsiders.com |
| Contents |
Rare elements on earth play an important role in today's life. They can be applied in scientific research to produce chemicals or other important substances. Learning functions and characteristics of rare elements wil help us have insights about science.
What are rare earth elements (REEs)?
A greener economy requires a certain set of metals known as rare earth elements, and finding the reserves for mining them is becoming more and more valuable.
The majority of rare earth elements are employed in conventional and low-carbon technologies as magnets and catalysts. Other significant applications for rare earth elements include the manufacture of glass, high-performance electronics, and unique metal alloys.
| With estimated rare earth oxide reserves and resources (measured and indicated) of over 14 million tonnes in 2021, Canada is one of the world's leading rare earth producers. The production of permanent magnets, which accounts for 29% of the total anticipated demand, is the primary global application for REEs. With an estimated 140,000 tonnes of REEs in 2020, or nearly 60% of global production, China is the world's largest producer. Rare earth resources are found in many nations, including Canada, but the production of REEs necessitates intricate separation and refining procedures. |
How are rare earth elements used?
Industrial applications for rare earth elements (REEs) include electronics, clean energy, aerospace, automotive, and defense.
The production of permanent magnets, which will account for 29% of the expected demand in 2020, is the single largest and most significant end use for REEs.
Modern electronics, found in wind turbines, jet aircraft, computers, televisions, cell phones, and many other products, depend on permanent magnets.
Because of their catalytic and luminescent qualities, REEs are also widely used in high-tech and "green" products.
REES are usedIn lights, screens, and glass: Rare earth elements (REEs) are used in electronics, clean energy, automotive, aerospace, and defense industries. The single largest and most important end use for REEs is the production of permanent magnets, which will make up 29% of the expected demand in 2020. Permanent magnets are essential to modern electronics, which are found in many products such as cell phones, computers, jet aircraft, wind turbines, and televisions. REEs are also extensively utilized in high-tech and "green" products due to their catalytic and luminescent properties. As catalysts: "Lanthanum-based catalysts are used to refine petroleum. Cerium-based catalysts are used in automotive catalytic converters" In magnets: The use of REE-based magnets is expanding quickly. The strongest magnets currently available are neodymium-iron-boron magnets, which are advantageous when weight and space are constraints. Hard drives in computers, as well as CD-ROM and DVD disk drives, employ rare-earth magnets. A rare-earth magnet drives a disk drive's spindle to a high degree of spinning stability. Several other common automotive subsystems, including audio speakers, power seats, electric windows, and power steering, also make use of these magnets. In batteries: Anodes for nickel-metal hydride batteries are made of alloys based on lanthanum. These battery types, which can require up to 10 to 15 kilograms of lanthanum per electric vehicle, are utilized in hybrid electric cars. In steel alloys: In the process of making steel, cerium, lanthanum, neodymium, and praseodymium—often in the form of a mixed oxide called mischmetal—are used to eliminate impurities and to create unique alloys. |
Top 11 Rarest Elements On Earth Today
1. Astatine
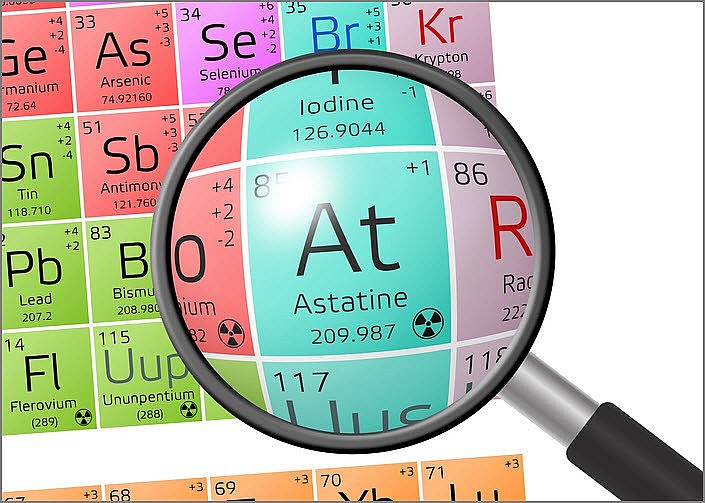 |
| Photo Thermo Fisher |
Abundance: less than 1 gram present in Earth’s crust at any given time; only 0.05 micrograms have ever been produced
Atomic Number: 85
Atomic Weight: 210 (most stable isotope)
Element Category: Metalloid
Common Uses: Currently being researched for use in nuclear medicine; has potential for targeted alpha-particle therapy.
Astatine is the rarest element on Earth, with only about 25 grams naturally occurring on the planet at any given time. Its existence was predicted in the 1800s, but it wasn't discovered until about 70 years later. Despite its discovery decades ago, very little is known about astatine. Indeed, physicists infer many of its properties, including radioactivity, conduction, and color, from other halogen group members.
2. Oganesson
Abundance: few milligrams produced as byproduct in nuclear reactors; just over 1 gram ever produced in the United States since it was first discovered
Atomic Number: 118
Atomic Weight: 294 (most stable isotope)
Element Category: Unknown chemical properties; possibly a metallic-looking reactive solid
Common Uses: No practical uses outside of scientific research
Oganesson is a radioactive, man-made element about which little is known. It is classified as a non-metal and is expected to be a gas. It belongs to the noble gas family.
The element, which is numbered 118 on the Periodic Table of Elements, was previously known as ununoctium, a placeholder name that means "one-one-eight" in Latin. The International Union of Pure and Applied Chemistry (IUPAC) approved the name oganesson for element 118 in November 2016.
| IUPAC officials said the name oganesson honors Yuri Oganessian "for his pioneering contributions to transactinide elements research," referring to elements with atomic numbers 104 through 120. "Among his numerous accomplishments are the discovery of super-heavy elements and significant advances in the nuclear physics of super-heavy nuclei, including experimental evidence for the 'island of stability,'" an idea that super-heavy elements can become stable at some point in their existence. |
3. Berkelium
 |
| Photo livescience |
Abundance: few milligrams produced as byproduct in nuclear reactors; just over 1 gram ever produced in the United States since it was first discovered
Atomic Number: 97
Atomic Weight: 247 (most stable isotope)
Element Category: Actinide
Common Uses: No practical uses outside of scientific research
Berkelium, a heavy metal element discovered by researchers, has an unusual property that could be used to distinguish it from other radioactive actinides. Scientists at the Department of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) discovered that the element berkelium breaks form with its heavy element peers when bound to a synthetic organic molecule using a variety of spectroscopic techniques. This property may aid scientists in developing improved methods for handling and purifying nuclear materials.
4. Francium
Abundance: between 20 to 30 grams at any given time found in Earth’s crust; a few hundred thousand atoms have been produced
Atomic Number: 87
Atomic Weight: 223 (most stable isotope)
Element Category: Alkali metal
Common Uses: No practical uses outside of scientific research in the fields of chemistry and of atomic structure.
Francium's characteristics:
- Francium is the least found metal on the planet earth and is rarely found in nature. It is considered to be the second rarest metal discovered on the earth’s crust next to the Astatine.
- Francium is an element with the chemical symbol Fr and atomic number 87 in the periodic table. It is produced both naturally and by artificial methods.
- Most probably, it is assumed that about 340-550 grams of this metal francium are found in the earth’s crust. Francium has a half-life of only 22 minutes.
- Francium occurs on the decay of the alpha particles, which are found in the minerals of uranium. Francium is obtained by the neutron bombardment of radium in a nuclear reactor. It can also be made by bombarding thorium with protons.
- This metal has about 34 isotopes that are said to be occurring in nature. Francium has only 1 valence electron.
| Since francium is produced in tiny quantities in nature, it does show any much commercial applications. Francium has been used in the field of research, chemistry and also in the atomic structure. It is used for diagnostics for curing cancers. It is also used in many spectroscopic experiments. Francium is a highly radioactive metal, and since it exhibits a short half-life, it does not have more impact on the environment. |
5. Protactinium
Abundance: few parts per trillion (0.1 part per trillion) in Earth’s crust; able to produce 125 grams at one time
Atomic Number: 91
Atomic Weight: 231.03588(1)
Element Category: Actinide
Common Uses: No practical uses outside of scientific research
Protactinium's characteristics:
- Protactinium is a radioactive element which belongs to the actinide series. It is dense and has a silver-grey appearance.
- The atomic mass of protactinium is 231.03.
- The melting point of protactinium is 1600°C.
- The boiling point of protactinium is unknown.
- The density of protactinium is 15370 in S.I. units at 20°C.
- Protactinium crystallizes in body-centered tetragonal structure.
- The properties of protactinium are intermediate of thorium and uranium.
- Protactinium is paramagnetic in nature.
Health Effects on Exposure
When protactinium enters the body through inhalation or ingestion, it causes serious problems. Cancer is the most serious problem that it can cause. The movement of protactinium within the body has the potential to disrupt almost all organs.
Effects on Surroundings
Protactinium is not a major concern for environment.
6. Promethium
 |
| Photo wikicommons |
Abundance: only about 500 to 600 grams in Earth’s crust at any given time
Atomic Number: 61
Atomic Weight: 145 (most stable isotope)
Element Category: Lanthanide
Promethium-147 is mostly used in scientific research, but it is also used in luminous paint, atomic batteries, and thickness measurement devices.
Promethium, named after the Greek Titan who stole fire from Zeus and gave it to humanity, is a highly radioactive, rare earth element. It is found in the byproducts of uranium fission reactions and is not found anywhere else on Earth. Because of its scarcity, its primary purpose is research; it has applications in a variety of medical devices, batteries, and luminescent paint.
| Density: 4.17 ounces per cubic inch (7.22 grams per cubic cm) Phase at room temperature: solid Melting point: 2,088 degrees Fahrenheit (1,142 degrees Celsius) Boiling point: 5,972 F (3,300 C) Number of natural isotopes (atoms of the same element with a different number of neutrons): at least 38 radioactive isotopes Most common isotopes: Pm-145 (negligible percent of natural abundance), Pm-147 (negligible percent of natural abundance) |
7. Californium
Abundance: about 500 milligrams produced annually
Atomic Number: 98
Atomic Weight: 251 (most stable isotope)
Element Category: Actinide
Common Applications: Used to assist in the start-up of nuclear reactors; used in nuclear synthesis of higher mass elements; used in neutron moisture gauges; used as a neutron source to identify gold and silver ores via a technique known as neutron activation; neutrons used in the treatment of certain cervical and brain cancers; and many other practical applications.
Californium is a man-made, radioactive element that does not exist in nature. It's an actinide, one of the 15 radioactive metallic elements at the bottom of the periodic table. The pure metal is silvery-white, malleable, and soft enough to be cut with a razor blade. Californium has a moderate chemical reactivity. At room temperature, it slowly tarnishes — small pieces or foils of the metal begin to oxidize, but not violently.
| Density: Unknown Phase at room temperature: Solid Melting point: 1,652 F (900 C) Boiling point: Unknown Number of isotopes (atoms of the same element with a different number of neutrons): 20 isotopes whose half-lives are known with mass numbers 237 to 256. Most common isotopes: No naturally occurring isotopes |
8. Neptunium
 |
| Photo science photo library |
Abundance: only trace amounts found in the Earth’s crust; about 60,000 kilograms produced as a byproduct of nuclear power plants each year
Atomic Number: 93
Atomic Weight: 237 (most stable isotope)
Element Category: Actinide
Common Applications: Used as a precursor in the production of Plutonium; in devices for detecting high-energy (MeV) neutrons; and in radioisotope thermal generators to power spacecraft.
Neptunium was named after the planet Neptune because it is the solar system's next outermost planet after Uranus, and this element is the next one after uranium in the periodic table.
Edwin McMillan and Philip Abelson discovered neptunium in the United States in 1940. Scientists have discovered approximately 18 isotopes of neptunium. All of them are radioactive. Neptunium was once considered a very rare element, but it can now be produced relatively easily in a nuclear reactor. A nuclear reactor is a device that produces nuclear fission reactions. Nuclear fission is the process of splitting atoms caused by neutrons colliding with uranium or plutonium atoms. These collisions result in the formation of new elements. Neptunium is only used in specialized detection devices in the commercial world.
9. Americium
 |
| Photo wikicommons |
Abundance: possible trace amounts found in the Earth’s crust, but has never been confirmed; several kilograms produced each year
Atomic Number: 95
Atomic Weight: 243 (most stable isotope)
Element Category: Actinide
Common Applications: Ionization chamber smoke detectors, neutron sources, industrial gauges, and as a starting material for the production of other transuranic elements and transactinides.
According to Lenntech, americium occurs naturally in extremely trace amounts in uranium minerals as a result of nuclear reactions. Americium concentrations may have been higher in the past when local uranium concentrations were higher, resulting in more nuclear reactions.
According to the Royal Society of Chemistry, americium is primarily produced in nuclear reactors by bombarding plutonium with neutrons.
| Density: 7.91 ounces per cubic inch (13.69 grams per cubic cm) Phase at room temperature: solid Melting point: 2,149 degrees Fahrenheit (1,176 degrees Celsius) Boiling point: 3,652 F (2,011 C) Number of natural isotopes (atoms of the same element with a different number of neutrons): 0. There are at least 19 radioactive isotopes created in a lab. Most common isotopes: Am-241 (negligible percent of natural abundance), Am-243 (negligible percent of natural abundance) |
10. Curium
Abundance: Unknown if any trace amounts exist naturally; several kilograms produced each year
Atomic Number: 96
Atomic Weight: 247 (most stable isotope)
Element Category: Actinide
Common applications include the production of heavier actinides, the use of radionuclides as power sources in artificial pacemakers, and the use of alpha particle X-ray spectrometers.
Pierre and Marie Curie were the inspiration for the name. Curium is a silvery, hard, and brittle metal that tarnishes slowly in dry air at room temperature. Curium does not occur naturally; instead, it is typically created artificially in nuclear reactors via successive neutron captures by plutonium and americium isotopes.
Curium is a highly radioactive element that is more electropositive than aluminum and is chenically reactive. The fluorides are a class of curium compounds.
Although curium is listed after americium in the periodic table, it was discovered before americium and was the third transuranium element to be discovered. It was discovered in 1944 at the wartime Metallurgical Laboratory in Chicago by Glenn Seaborg, James, and Albert Ghiorso as a result of helium-ion bombardment of 239Pu in the Berkeley Radiation Laboratory's 60-inch cyclotron. Werner and Perlman of the University of California, Berkeley were the first to isolate visible amounts (30Mg) of 242Cm in the form of the hydroxide in 1947. Crane, Wallmann, and Cunningham discovered in 1950 that the magnetic susceptibility of microgram samples of CmF3 was the same as that of GdF3. This provided direct experimental evidence for accrediting Cm+3 with an electronic configuration. The same workers prepared curium in its elemental form for the first time in 1951.
| Curium-247, the longest-lived isotope of Curium, has a half-life of 15.6 million years, implying that any primordial Curium present on Earth during its formation would have already decayed. Curium traces have been discovered in areas used for atmospheric nuclear weapons tests. |
11. Tellurium
Abundance: present in the Earth's crust only in about 0.001 parts per million
Atomic Number: 52
Atomic Weight: 127.6 (most stable isotope)
Element Category: Metalloid
Common Uses: used to vulcanise rubber, to tint glass and ceramics, in solar cells, in rewritable CDs and DVDs and as a catalyst in oil refining. It can be doped with silver, gold, copper or tin in semiconductor applications.
Tellurium is a rare element that has metallurgical applications as an additive to stainless steel and as an ingredient in copper, lead, and iron alloys. According to the US Geological Survey Fact Sheet Tellurium —The Bright Future of Solar Energy, tellurium's primary use is in the manufacture of films required for thin film photovoltaic solar cells. Tellurium, when alloyed with other elements such as cadmium, forms a compound with increased electrical conductivity. A thin film can effectively absorb and convert sunlight into electricity.
 Top 10 Mysteries of Human Behaviour That Science Fails to Explain Top 10 Mysteries of Human Behaviour That Science Fails to Explain Human behaviour is an interesting topic to talk about, and scientists have been learning about it for years. Here is top 10 mysteries of human ... |
 How Old Is The Earth In Science, Bible How Old Is The Earth In Science, Bible Although the universe is thought to be about 13.77 billion years old, planet Earth is much younger than that. Scroll down to know how old ... |
 Religion and Science Are Both Necessary in Life Religion and Science Are Both Necessary in Life Both science and religion are necessary and indispensable in human life. |