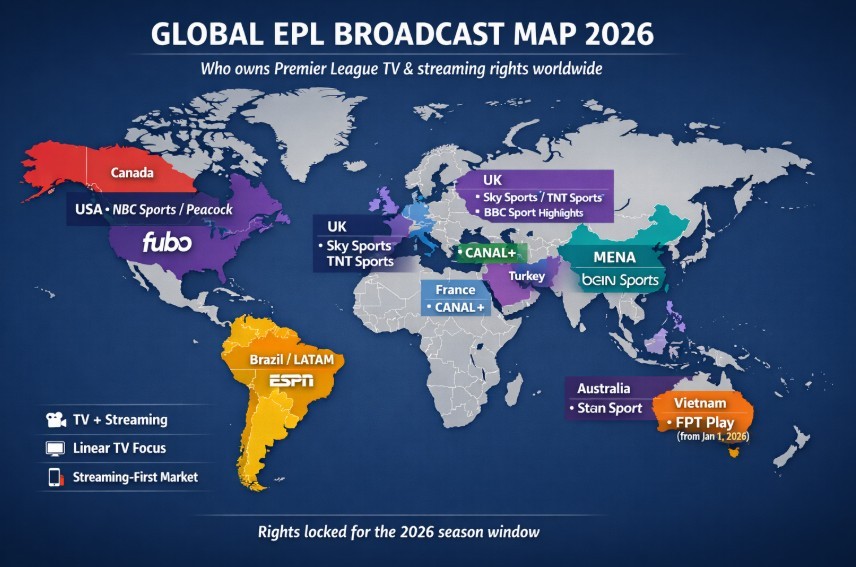
Famous Medical Device Failures: Lessons Learned and Ongoing Safety
 |
| The failure of a medical device can result if the loss of life |
| Contents |
However, the history of medical devices includes instances of high-profile failures that have had significant consequences for patients and the healthcare industry. This article will look into some famous medical device failures, the lessons learned from these incidents, and the ongoing efforts to enhance device safety.
1. DePuy Synthes Hip Implants (ASR and Pinnacle)
DePuy Synthes, a subsidiary of Johnson & Johnson, faced major controversy with its hip implants. The ASR (Articular Surface Replacement) and Pinnacle hip implants were associated with a higher-than-expected rate of device failure, leading to pain, metallosis (metal poisoning), and the need for revision surgeries. Thousands of patients filed lawsuits against the company, resulting in a multibillion-dollar settlement. This case underscored the importance of rigorous testing and monitoring of medical devices, particularly those with long-term implantation.
2. The Dalkon Shield IUD
In the 1970s, the Dalkon Shield intrauterine device (IUD) was marketed as a highly effective birth control option. However, it was later discovered that the device was associated with severe health risks, including pelvic inflammatory disease (PID), infertility, and even death. The Dalkon Shield IUD was eventually withdrawn from the market, highlighting the need for thorough pre-market testing and ongoing post-market surveillance of medical devices.
3. Medtronic Infuse Bone Graft
The Medtronic Infuse Bone Graft, used in spinal fusion surgeries, faced controversy when it was revealed that the device had been promoted for off-label use in ways not approved by the FDA. This led to concerns about complications such as uncontrolled bone growth and nerve damage. The case emphasized the importance of transparency in clinical research and the need for accurate reporting of adverse events associated with medical devices.
4. Boston Scientific's Riata Defibrillator Leads
The Riata defibrillator leads manufactured by Boston Scientific experienced issues with insulation integrity, leading to a risk of electrical malfunction. These problems raised concerns about the safety and reliability of implantable cardiac devices. The incident highlighted the need for comprehensive post-market surveillance, monitoring, and timely action to address potential device defects.
5. Therac-25 Radiation Therapy Machine
The Therac-25 radiation therapy machine was designed to treat cancer patients but had software and hardware flaws that resulted in massive overdoses of radiation. Several patients suffered severe injuries, and some died as a result. This catastrophic failure underscored the critical importance of rigorous software testing and safety checks in medical devices, particularly those involving radiation therapy.
Lessons Learned and Ongoing Safety Efforts
These famous medical device failures serve as stark reminders of the importance of patient safety and rigorous regulatory oversight. Lessons learned from these incidents have led to significant improvements in the medical device industry:
- Regulatory Enhancements: regulatory agencies like the FDA have increased scrutiny of medical devices, implementing more rigorous pre-market evaluations and post-market surveillance.
- Adverse Event Reporting: mandatory reporting of adverse events and complications associated with medical devices has become more stringent, ensuring that potential issues are identified and addressed promptly.
- Transparency: transparency in clinical trials and device testing is now emphasized, allowing healthcare professionals and patients to make informed decisions.
- Patient Advocacy: patients and patient advocacy groups play an essential role in raising awareness of device safety issues and advocating for improvements in device regulation and oversight. Professionals, such as a defective medical device lawyer, bring some power back to patients.
- Research and Development: medical device companies invest heavily in research and development, incorporating advanced testing and quality control measures to enhance device safety.
- Education and Training: healthcare professionals receive improved education and training on the proper use of medical devices, ensuring that they’re used safely and effectively.
While the history of medical devices includes notable failures, it also demonstrates the capacity of the healthcare industry to learn from mistakes and continually improve device safety. Patients and healthcare providers can contribute to ongoing safety efforts by reporting adverse events and advocating for the highest standards of device safety and efficacy. In this way, the healthcare community can strive to prevent future high-profile medical device failures and prioritize patient well-being.
 10+ Best Free Online Courses for Medical Billing and Coding Today 10+ Best Free Online Courses for Medical Billing and Coding Today You should find out about the different kinds of training that are offered if you're thinking about a career in medical billing and coding.The top ... |
 How To Remove Acne Scars With The Natural & Medical Treatments How To Remove Acne Scars With The Natural & Medical Treatments There are several tips to get rid of acne scars that you can do at home rignt now. |
 Top 10+ Most Prestigious Premedical Colleges In The US Today Top 10+ Most Prestigious Premedical Colleges In The US Today Are you looking for a prestigious school to study premedical? Check out this article to know Top 10+ Most Prestigious Premedical Colleges In The US ... |